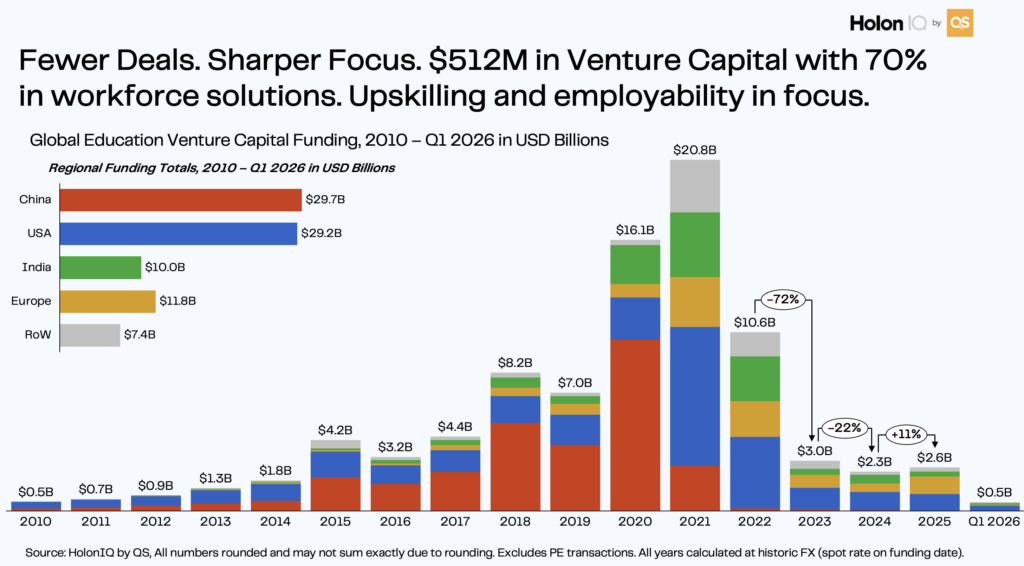
Biological Superintelligence: Owkin and AstraZeneca’s $4.7B Bet on AI Scientists
Owkin and AstraZeneca’s agentic AI drug discovery 2026 partnership is deploying autonomous AI scientists to accelerate cancer cures. Discover how biological superintelligence is reshaping pharmaceutical R&D.
Introduction: The Scientist That Never Sleeps
Imagine a research scientist who never needs coffee, never requests vacation, and can read every paper ever published on a disease in an afternoon. Now imagine that scientist does not just retrieve information—it reasons. It forms hypotheses. It designs experiments. It learns from its mistakes and adjusts its strategy without human prompting.
That is not a future fantasy. That is agentic AI—and it is coming to a laboratory near you.
On May 13, 2026, Owkin, the agentic AI company pioneering Biological Artificial Superintelligence, finalized a landmark agreement with AstraZeneca. The deal, structured as a three‑year licensing of Owkin’s AI Scientist platform K Pro, will see Owkin build specialized biopharma agents capable of high‑level strategic decision‑making in drug discovery .
This is not a typical software license. It is not a chatbot. It is a fundamental shift in how pharmaceutical research operates—moving AI from the role of assistant to the role of principal investigator.
In this article, we will explore the agentic AI drug discovery 2026 revolution, the technology powering Owkin’s “biological superintelligence,” and why this $4.7 billion bet may reshape the timeline for finding cures.
Part 1: The Deal – What Owkin and AstraZeneca Are Building
1.1 K Pro: The AI Scientist
At the heart of the agreement is K Pro—Owkin’s “AI Scientist for biopharmaceutical research and strategic decision‑making” . Unlike traditional AI models that generate text or classify images, K Pro is an agentic system: a suite of AI skills and tools that can orchestrate complex workflows, decode multimodal biological data, and take autonomous action within established governance frameworks.
The three‑year licensing agreement gives AstraZeneca access to K Pro’s full capabilities. But the more significant component is this: Owkin will develop novel biopharma agents specifically for AstraZeneca, integrated directly into the company’s IT infrastructure and decision workflows .
These agents are not generic. They are purpose‑built for competitive intelligence—analyzing pharmaceutical targets, forecasting the landscape for specific assets, and providing executives with timely, data‑rich insights that previously required weeks of manual analysis .
1.2 From Diagnostics to Superintelligence
The partnership did not emerge from nowhere. It builds on a successful collaboration between Owkin and AstraZeneca to develop an AI‑powered gBRCA pre‑screen solution for breast cancer. At ESMO, the BRCAura RUO solution demonstrated the ability to rule out approximately 40% of patients unlikely to carry gBRCA mutations with a 93% sensitivity . That work now continues at Waiv, Owkin’s spun‑out diagnostics division, while Owkin doubles down on its core mission: biological artificial superintelligence .
Thomas Clozel, MD, co‑founder and CEO of Owkin, put it simply: “At Owkin, we believe the future of the pharmaceutical industry is agentic. Our experience, multimodal data, and agentic infrastructure allows us to build various complex agents supporting our pharmaceutical partners, including competitive intelligence agents to support quick decisions by executives” .
1.3 The Financial Scope
While the exact valuation of the AstraZeneca agreement has not been publicly disclosed, the broader context is clear. Owkin has raised over $300 million from leading biopharma companies including Sanofi and BMS, as well as venture funds like F‑Prime, GV, and Bpifrance . The company is actively developing frontier models for biological reasoning, accelerated by a collaboration with NVIDIA announced in January 2026 .
Owkin’s ambition is nothing less than the world’s first Biological Artificial Super Intelligence (BASI) —an AI system capable of understanding and reasoning about the complexity of biology at a scale previously impossible .
Part 2: What Is Agentic AI, and Why Does It Matter for Drug Discovery?
2.1 Beyond Chatbots: The Autonomous Researcher
To understand why this matters, we must distinguish between three levels of AI capability:
| Level | Capability | Example |
|---|---|---|
| Assistive AI | Responds to queries, retrieves information | ChatGPT, standard LLMs |
| Copilot AI | Suggests actions, drafts content | Github Copilot, Owkin K |
| Agentic AI | Takes autonomous action, orchestrates workflows, revises strategies based on outcomes | K Pro agents, autonomous hypothesis testing |
Agentic AI is defined by autonomy. Given a high‑level goal—”analyze the competitive landscape for this drug target”—an agentic system can break the goal into sub‑tasks, decide which data sources to query, execute analyses, and present findings without step‑by‑step human guidance .
2.2 The Four Capabilities of K Pro
According to the announcement, K Pro’s agentic infrastructure provides four core capabilities for biopharma decision‑making :
- Multimodal data integration: The system ingests not just text, but genomics, pathology images, clinical trial data, and real‑world patient records.
- Competitive intelligence automation: Agents can rapidly analyze and forecast the landscape for specific pharmaceutical targets, assets, and trials.
- End‑to‑end development: Owkin leads the full development of agents, from design through deployment within AstraZeneca’s secure infrastructure.
- Governance and security: All agents operate within established enterprise standards—no black boxes, no uncontrolled data access.
2.3 Closing the Wet‑Lab/Data Science Gap
One of the persistent inefficiencies in drug discovery is the gap between wet‑lab scientists (who run experiments) and data scientists (who build models). The two groups speak different languages, use different tools, and often work in isolation .
Agentic AI bridges this gap. As Dr. Natalie van Zuydam, Executive Director of Data Sciences at AstraZeneca, explained, “Agentic AI sits across these layers and removes friction. Pair a model like AlphaFold with a natural‑language agent, and a wet‑lab scientist can run complex workflows without deep data science expertise. The barrier drops. Usage climbs. Feedback loops tighten” .
The result? Smaller, more focused studies that move projects faster through the pipeline.
Part 3: The Technology Behind Biological Superintelligence
3.1 OwkinZero and NVIDIA Collaboration
Owkin’s biological reasoning capabilities are powered by OwkinZero, a frontier large reasoning model fine‑tuned specifically for biology. In January 2026, Owkin announced a collaboration with NVIDIA to accelerate OwkinZero’s development, leveraging NVIDIA’s Nemotron family of open models and the NeMo framework for reinforcement learning .
The NVIDIA collaboration focuses on improving performance, scalability, guardrails, and robustness—critical requirements for deploying AI in regulated pharmaceutical environments .
3.2 Multimodal Patient Data
What distinguishes Owkin from general‑purpose AI companies is its access to unrivalled multimodal patient data. Through partnerships with leading cancer centers like Gustave Roussy (France) and Charité Comprehensive Cancer Center (Germany), Owkin has built a pan‑European agentic infrastructure for making biological data AI‑ready .
This infrastructure, unveiled at the Franco‑German Summit on Digital Sovereignty in November 2025, is designed to harmonize and structure biomedical data across Europe, powering a new reasoning model capable of automating every stage of biological research .
3.3 Federated Learning and Open Source
Critically, Owkin’s technology respects data privacy. The company’s federated learning software, Substra, allows AI models to learn from distributed datasets without any data leaving its original location. In the MELLODDY project, ten competing pharmaceutical companies—including Amgen, AstraZeneca, Bayer, GSK, Novartis, and Merck—agreed to share drug discovery data through this privacy‑preserving architecture .
In March 2026, Owkin placed Substra under the stewardship of the Linux Foundation, making it open source. As Ibrahim Haddad, General Manager of the LF AI & Data Foundation, stated: “Innovation thrives in collaboration, not in isolation” .
Part 4: The Broader Context – The “Prompt‑to‑Drug” Revolution
Owkin and AstraZeneca are not alone in racing toward autonomous drug discovery. In February 2026, researchers from Insilico Medicine and Eli Lilly published a landmark perspective in ACS Central Science describing a framework for fully autonomous, AI‑orchestrated drug discovery .
The vision, called “prompt‑to‑drug,” imagines a scientist simply requesting, “Design a drug for idiopathic pulmonary fibrosis.” A central AI controller would autonomously delegate tasks across target discovery, generative chemistry, automated synthesis, biological validation, and clinical planning .
While that future remains aspirational, the foundational components are already operational. Insilico has already reduced preclinical development timelines from 3–6 years to just 12–18 months per program, synthesizing only 60–200 molecules per candidate .
Part 5: Why This Matters – AI Leading, Not Assisting
5.1 Speed to Cure
The most immediate impact of agentic AI in drug discovery is speed. Traditional pharmaceutical R&D is slow because it is serial: hypothesis, experiment, analyze, repeat. Agentic AI can parallelize these steps, test multiple hypotheses simultaneously, and learn from every outcome in real time.
For cancer patients waiting for new therapies, every month matters. Agentic AI promises to cut years off the search for cures.
5.2 Hypothesis Generation at Scale
Human researchers, no matter how brilliant, suffer from cognitive biases and blind spots. We tend to pursue the hypotheses that are familiar, fundable, or fashionable. Agentic AI has no such constraints. It can explore the full space of biological possibility, generating novel hypotheses that no human would have considered.
5.3 The “Superintelligence” Threshold
Owkin’s ultimate goal is Biological Artificial Superintelligence (BASI) —an AI system that understands complex biology at a level beyond any human researcher . This does not mean the AI replaces humans. It means the AI becomes a partner capable of identifying patterns, connections, and mechanisms that human researchers, working alone, would likely miss.
As Europe’s Franco‑German digital sovereignty initiative noted: “Europe can be number one in biological AI. While the race for general‑purpose LLMs has largely been won by American companies, the field of biology‑native reasoning systems remains wide open” .
Part 6: Challenges and Caveats
6.1 Hallucination and Error Propagation
Agentic AI systems can “hallucinate”—generating plausible‑sounding but incorrect outputs. In drug discovery, a hallucinated protein structure or misread genomic variant could send research down costly dead ends. Safeguards, auditability, and human oversight for high‑stakes decisions remain essential .
6.2 Regulatory Uncertainty
How do you validate an AI‑generated hypothesis? How do you patent a molecule discovered by an autonomous agent? Regulatory frameworks for AI‑driven drug discovery are still evolving. The Insilico/Lilly paper recommends the use of “AI arms” in clinical trials—comparing AI‑driven predictions against traditional methods in real‑world settings—to validate the technology .
6.3 Data Privacy and Sovereignty
Owkin’s federated learning approach addresses many privacy concerns, but the fundamental tension remains: AI models need data to learn, and patient data is sensitive. The pan‑European infrastructure being built by Owkin, Gustave Roussy, and Charité is designed to meet the highest standards of data protection while enabling collaboration .
Conclusion: The Principal Investigator Is an Algorithm
The agentic AI drug discovery 2026 partnership between Owkin and AstraZeneca marks a turning point. For the first time, an AI system will sit alongside human executives, providing competitive intelligence, forecasting trial outcomes, and making high‑level decisions without step‑by‑step prompting.
This is not the end of human researchers. It is the elevation of their role. The tedious work of literature review, data aggregation, and preliminary hypothesis testing will be automated. Scientists will be free to focus on what only humans can do: asking the bold questions, designing the creative experiments, and interpreting the AI’s findings with wisdom and skepticism.
The AI scientist does not sleep. It does not forget. It does not get bored.
But it still needs a partner who knows what questions are worth asking.
Frequently Asked Questions (FAQ)
What is Owkin’s K Pro?
K Pro is Owkin’s “AI Scientist”—an agentic AI platform that orchestrates a suite of AI skills and tools to decode complex biology, accelerate pharmaceutical research, and support strategic decision‑making .
How does agentic AI differ from ChatGPT?
ChatGPT is assistive—it responds to prompts. Agentic AI can take autonomous action, break goals into sub‑tasks, orchestrate workflows, and revise strategies based on outcomes without continuous human guidance .
What is the financial scope of the Owkin-AstraZeneca deal?
The agreement is a three‑year licensing of K Pro, including custom biopharma agent development. While the exact value is undisclosed, the deal represents a major bet on agentic AI as the future of pharmaceutical R&D.
What is Biological Artificial Superintelligence (BASI)?
BASI is Owkin’s term for an AI system capable of understanding and reasoning about the complexity of biology at a scale beyond human researchers, accelerating the discovery of new cures and diagnostics .
Is agentic AI already used in drug discovery?
Yes. The K Pro platform is currently in use, and Insilico Medicine has already reduced preclinical timelines from 3–6 years to 12–18 months using AI‑driven workflows. The Owkin-AstraZeneca agreement moves agentic AI from discovery support into strategic decision‑making .
Call to Action (CTA)
Are you a researcher, pharmaceutical executive, or student interested in the future of AI‑driven drug discovery? Share your perspective in the comments below. And if you found this article valuable, share it with a colleague who cares about how technology is transforming the fight against cancer.






